Sodium facts for kids
 |
||||||||||||||||||||||||||||
| Sodium | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Appearance | silvery white metallic | |||||||||||||||||||||||||||
| Standard atomic weight Ar, std(Na) | 22.98976928(2) | |||||||||||||||||||||||||||
| Sodium in the periodic table | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Atomic number (Z) | 11 | |||||||||||||||||||||||||||
| Group | group 1: hydrogen and alkali metals | |||||||||||||||||||||||||||
| Period | period 3 | |||||||||||||||||||||||||||
| Block | s | |||||||||||||||||||||||||||
| Electron configuration | [Ne] 3s1 | |||||||||||||||||||||||||||
| Electrons per shell | 2,8,1 | |||||||||||||||||||||||||||
| Physical properties | ||||||||||||||||||||||||||||
| Phase at STP | solid | |||||||||||||||||||||||||||
| Melting point | 370.87 K (97.72 °C, 207.9 °F) | |||||||||||||||||||||||||||
| Boiling point | 1156 K (883 °C, 1621 °F) | |||||||||||||||||||||||||||
| Density (near r.t.) | 0.968 g/cm3 | |||||||||||||||||||||||||||
| when liquid (at m.p.) | 0.927 g/cm3 | |||||||||||||||||||||||||||
| Critical point | (extrapolated) 2573 K, 35 MPa |
|||||||||||||||||||||||||||
| Heat of fusion | 2.60 kJ/mol | |||||||||||||||||||||||||||
| Heat of vaporization | 97.42 kJ/mol | |||||||||||||||||||||||||||
| Molar heat capacity | 28.230 J/(mol·K) | |||||||||||||||||||||||||||
Vapor pressure
|
||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||
| Oxidation states | −1, +1 (a strongly basic oxide) | |||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 0.93 | |||||||||||||||||||||||||||
| Ionization energies |
|
|||||||||||||||||||||||||||
| Atomic radius | empirical: 186 pm | |||||||||||||||||||||||||||
| Covalent radius | 166±9 pm | |||||||||||||||||||||||||||
| Van der Waals radius | 227 pm | |||||||||||||||||||||||||||
| Spectral lines of Sodium | ||||||||||||||||||||||||||||
| Other properties | ||||||||||||||||||||||||||||
| Natural occurrence | primordial | |||||||||||||||||||||||||||
| Crystal structure | body-centered cubic (bcc) | |||||||||||||||||||||||||||
| Speed of sound thin rod | 3200 m/s (at 20 °C) | |||||||||||||||||||||||||||
| Thermal expansion | 71 µm/(m⋅K) (at 25 °C) | |||||||||||||||||||||||||||
| Thermal conductivity | 142 W/(m⋅K) | |||||||||||||||||||||||||||
| Electrical resistivity | 47.7 n Ω⋅m (at 20 °C) | |||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic | |||||||||||||||||||||||||||
| Young's modulus | 10 GPa | |||||||||||||||||||||||||||
| Shear modulus | 3.3 GPa | |||||||||||||||||||||||||||
| Bulk modulus | 6.3 GPa | |||||||||||||||||||||||||||
| Mohs hardness | 0.5 | |||||||||||||||||||||||||||
| Brinell hardness | 0.69 MPa | |||||||||||||||||||||||||||
| CAS Number | 7440-23-5 | |||||||||||||||||||||||||||
| History | ||||||||||||||||||||||||||||
| Discovery | Humphry Davy (1807) | |||||||||||||||||||||||||||
| First isolation | Humphry Davy (1807) | |||||||||||||||||||||||||||
| Symbol | "Na": from New Latin natrium, coined from German Natron, 'natron' | |||||||||||||||||||||||||||
| Main isotopes of Sodium | ||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
Sodium is a chemical element with the symbol Na (from Latin natrium) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table, because it has a single electron in its outer shell, which it readily donates, creating a positively charged ion—the Na+ cation. Its only stable isotope is Na. The free metal does not occur in nature, and must be prepared from compounds. Sodium is the sixth most abundant element in the Earth's crust and exists in numerous minerals such as feldspars, sodalite, and rock salt (NaCl). Many salts of sodium are highly water-soluble: sodium ions have been leached by the action of water from the Earth's minerals over eons, and thus sodium and chlorine are the most common dissolved elements by weight in the oceans.
Sodium was first isolated by Humphry Davy in 1807 by the electrolysis of sodium hydroxide. Among many other useful sodium compounds, sodium hydroxide (lye) is used in soap manufacture, and sodium chloride (edible salt) is a de-icing agent and a nutrient for animals including humans.
Sodium is an essential element for all animals and some plants. Sodium ions are the major cation in the extracellular fluid (ECF) and as such are the major contributor to the ECF osmotic pressure and ECF compartment volume. Loss of water from the ECF compartment increases the sodium concentration, a condition called hypernatremia. Isotonic loss of water and sodium from the ECF compartment decreases the size of that compartment in a condition called ECF hypovolemia.
Contents
Properties
Sodium is a light, silver-coloured metal. Sodium is so soft that it can be easily cut with a knife. When it is cut, the surface will become white after a bit of time. This is because it reacts with air to form sodium hydroxide and sodium carbonate. Sodium is a little lighter than water; when it reacts with water it floats. This reaction is very fast. Hydrogen and sodium hydroxide are produced. The hydrogen may ignite. Since sodium melts at a low temperature, it melts when it reacts with water. It has one valence electron which is removed easily, making it highly reactive.
Compared with other alkali metals (metals in the first column of the periodic table), sodium is usually less reactive than potassium and more reactive than lithium.
Chemical compounds
These are chemical compounds that contain sodium ions. Sodium only exists in 1 oxidation state: +1.
- Sodium aluminum fluoride, used to make aluminum
- Sodium amide, very strong base
- Sodium arsenite, colorless solid, very toxic
- Sodium arsenate, oxidizing agent, very toxic
- Sodium azide, used in airbags
- Sodium bicarbonate, baking soda, used in cooking
- Sodium bismuthate, oxidizing agent, used to test for manganese
- Sodium bisulfate, acidic, used to increase pH
- Sodium bromate, oxidizing agent, used to dye hair
- Sodium bromide, rare, used in some medicine
- Sodium carbonate, used to make glass
- Sodium chlorate, used in some explosives
- Sodium chlorite, used in disinfectants
- Sodium chloride, table salt
- Sodium chromate, yellow, oxidizing agent, toxic
- Sodium dichromate, orange, oxidizing agent, toxic
- Sodium fluoride, used in toothpastes, bitter, toxic in large doses
- Sodium hydroxide, lye, used in soap, strong base
- Sodium hypochlorite, bleach, disinfectant
- Sodium hypophosphite, reducing agent, poisonous
- Sodium iodate, oxidizing agent, prevents iodine deficiency
- Sodium iodide, weak reducing agent, prevents iodine deficiency
- Sodium manganate, rare green solid
- Sodium nitrate, used in blasting powder
- Sodium nitrite, used in food preservation
- Sodium periodate, oxidizing agent
- Sodium permanganate, less common than potassium permanganate, oxidizing agent
- Sodium phosphate, various uses
- Sodium phosphide, catalyst
- Sodium phosphite, toxic, reducing agent
- Sodium selenate, strong oxidizing agent, other selenium compounds
- Sodium selenide, strong reducing agent, reactive
- Sodium selenite, weak oxidizing agent, vitamin supplement
- Sodium sulfate, bitter, laxative
- Sodium sulfite, weak reducing agent, used to preserve dried food
- Sodium tellurate, strong oxidizing agent
- Sodium telluride, strong reducing agent, reacts with air easily
- Sodium tellurite, main tellurite compound
Discovery and name
Sodium was discovered by Sir Humphrey Davy, an English scientist, back in 1807. He made it by the electrolysis of sodium hydroxide. It is named after soda, a name for sodium hydroxide or sodium carbonate.
Use as element
It is used in the preparation of organic compounds. It is also used in the street lights that are orange, and ultra violet lights.
Use as compounds
Sodium compounds are used in soaps, toothpaste, baking and antiacids. .
Occurrence and production
Sodium does not exist as an element in nature; its easily removed valence electron is too reactive. It exists as an ion in chemical compounds. Sodium ions are found in the ocean. It is also found as sodium chloride in the earth's crust, where it is mined.
Sodium is normally made by electrolysis of very hot sodium chloride that was melted.
Use in organisms
Sodium ion in the form of sodium chloride is needed in the human body, but large amounts of it cause problems, which is why one should not eat too much salt. Many organisms in the ocean depend on the proper concentration of ions in sea water to live.
Related pages
- List of common elements
- Hyponatremia (a medical problem caused by not having enough sodium in the body)
Images for kids
-
Emission spectrum for sodium, showing the D line.
-
A positive flame test for sodium has a bright yellow color.
-
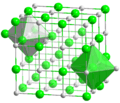
The structure of sodium chloride, showing octahedral coordination around Na+ and Cl− centres. This framework disintegrates when dissolved in water and reassembles when the water evaporates.
See also
 In Spanish: Sodio para niños
In Spanish: Sodio para niños