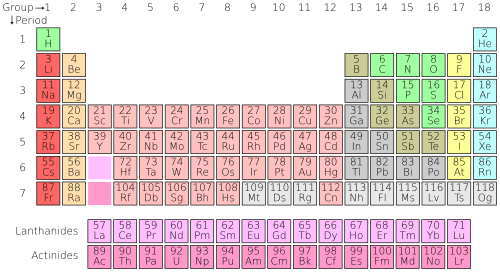Periodic table facts for kids
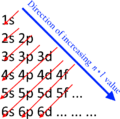
The table of chemical elements is a list of known chemical elements. As of 2022[update], a total of 118 elements have been discovered and confirmed. In the table, the elements are placed in the order of their atomic numbers starting with the lowest number of one, hydrogen. The atomic number of an element is the same as the number of protons in that particular nucleus of an atom. In the periodic table the elements are arranged into periods and groups. A row of elements across the table is called a period. Each period has a number; from 1 to 8. Period 1 has only 2 elements in it: hydrogen and helium. Period 2 and Period 3 both have 8 elements. Other periods are longer. Elements in a period have consecutive atomic numbers.
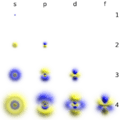
A column of elements down the table is called a group. There are 18 groups in the standard periodic table. Each group has a number: from 1 to 18. Elements in a group have electrons arranged in similar ways, according to the number of valency electrons, which gives them similar chemical properties (they behave in similar ways). For example, group 18 is known as the noble gases because they are all gases and they do not combine with other atoms.
There are two systems of group numbers; one using Arabic numerals (1,2,3) and the other using Roman numerals (I, II, III). The Roman numeral names were used in most of the 20th century. In 1990 the International Union of Pure and Applied Chemistry (IUPAC) decided to use the new system with Arabic numerals, to replace the two old group systems that used Roman numerals.
The periodic table has been used by chemists to observe patterns and relationships between elements. There are 3 main groups in the Periodic Table; metals, metalloids, and nonmetals. For example, elements to the bottom and far left of the table are the most metallic, and elements on the top right are the least metallic. (e.g. cesium is much more metallic than helium). There are also many other patterns and relationships.
The periodic table was invented by the Russian chemist Dmitry Ivanovich Mendeleyev (1834–1907). In his honor, element 101 was named after him, mendelevium.
Contents
Standard periodic table
| Group → | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | ||
| Period ↓ | ||||||||||||||||||||
| 1 | 1 H |
2 He |
||||||||||||||||||
| 2 | 3 Li |
4 Be |
5 B |
6 C |
7 N |
8 O |
9 F |
10 Ne |
||||||||||||
| 3 | 11 Na |
12 Mg |
13 Al |
14 Si |
15 P |
16 S |
17 Cl |
18 Ar |
||||||||||||
| 4 | 19 K |
20 Ca |
21 Sc |
22 Ti |
23 V |
24 Cr |
25 Mn |
26 Fe |
27 Co |
28 Ni |
29 Cu |
30 Zn |
31 Ga |
32 Ge |
33 As |
34 Se |
35 Br |
36 Kr |
||
| 5 | 37 Rb |
38 Sr |
39 Y |
40 Zr |
41 Nb |
42 Mo |
43 Tc |
44 Ru |
45 Rh |
46 Pd |
47 Ag |
48 Cd |
49 In |
50 Sn |
51 Sb |
52 Te |
53 I |
54 Xe |
||
| 6 | 55 Cs |
56 Ba |
Lanthanides | 72 Hf |
73 Ta |
74 W |
75 Re |
76 Os |
77 Ir |
78 Pt |
79 Au |
80 Hg |
81 Tl |
82 Pb |
83 Bi |
84 Po |
85 At |
86 Rn |
||
| 7 | 87 Fr |
88 Ra |
*Actinides |
104 Rf |
105 Db |
106 Sg |
107 Bh |
108 Hs |
109 Mt |
110 Ds |
111 Rg |
112 Cn |
113 Nh |
114 Fl |
115 Mc |
116 Lv |
117 Ts |
118 Og |
||
| 8 | 119 Uue |
120 Ubn |
**Superactinides |
158 Upo |
159 Upo |
160 Uhn |
161 Uhu |
162 Uhb |
163 Uht |
164 Uhq |
165 Uhp |
166 Uhh |
167 Uhs |
168 Uho |
169 Uhe |
170 Usn |
171 Usu |
172 Usb |
||
| 9 | 173 Ust |
174 Usq |
||||||||||||||||||
| * Lanthanide Series | 57 La |
58 Ce |
59 Pr |
60 Nd |
61 Pm |
62 Sm |
63 Eu |
64 Gd |
65 Tb |
66 Dy |
67 Ho |
68 Er |
69 Tm |
70 Yb |
71 Lu |
|||||
| ** Actinide Series | 89 Ac |
90 Th |
91 Pa |
92 U |
93 Np |
94 Pu |
95 Am |
96 Cm |
97 Bk |
98 Cf |
99 Es |
100 Fm |
101 Md |
102 No |
103 Lr |
|||||
| *** Superactinide Series | 143 Uqt |
144 Uqq |
145 Uqp |
146 Uqh |
147 Uqs |
148 Uqo |
149 Uqe |
150 Upn |
151 Upu |
152 Upb |
153 Upt |
154 Upq |
155 Upp |
156 Uph |
157 Ups |
|||||
| *** Superactinide Series | 121 Ubu |
122 Ubb |
123 Ubt |
124 Ubq |
125 Ubp |
126 Ubh |
127 Ubs |
128 Ubo |
129 Ube |
130 Utn |
131 Utu |
132 Utb |
133 Utt |
134 Utq |
135 Utp |
136 Uth |
137 Uts |
138 Uto |
139 Ute |
140 Uqn |
141 Uqu |
142 Uqb |
- Chemical Series of the Periodic Table
-
Superactinides
- State at standard temperature and pressure. The color of the number (atomic number) above the element symbol shows the state of the element at normal conditions.
- those in blue are gases
- those in green are liquids
- those in black are solid
-
Those with solid borders have stable isotopes (Primordial elements)
-
Those with dashed borders have only radioactive naturally occurring isotopes
-
Those with dotted borders do not occur naturally (Synthetic Elements)
-
those without borders are too radioactive to have been discovered yet.
Other methods for displaying the chemical elements
The version of the periodic table shown above is the one most used. Other widespread versions are shown below:
-
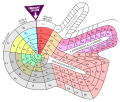
Theodor Benfey arranged the elements in a spiral, around hydrogen. The atomic weight determines the position of the element.
-
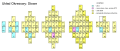
Dmitry Ivanovich Mendeleyev used a flower arrangement; Actinides, Lanthanides are shown as loops beside the main group.
-
Betterman arranged the elements by their isoelectric properties, which can be converted to a polynomial form.
Versions used less
- The standard table provides the basics. It is shown above
- A vertical table for improved readability in web browsers
- The big table provides the basics plus full element names
- The huge table provides the basics plus full element names and atomic masses
- Electron configurations
- Metals and Non Metals
- List of elements: includes name, symbol, atomic number, atomic mass, group, and period; sortable by any of those
- List of elements by symbol
- List of elements by boiling point
- List of elements by melting point
- List of elements by density
Related pages
Images for kids
See also
 In Spanish: Tabla periódica de los elementos para niños
In Spanish: Tabla periódica de los elementos para niños