Ammonium chloride facts for kids
Quick facts for kids Ammonium chloride |
|
|---|---|
 |
|
 |
|
| IUPAC name | Ammonium chloride |
| Other names | Sal ammoniac, Salmiac, Nushadir salt, Sal armagnac, Salt armoniack, Salmiak |
| Identifiers | |
| CAS number | |
| PubChem | |
| EC number | 235-186-4 |
| KEGG | D01139 |
| ChEBI | CHEBI:31206 |
| RTECS number | BP4550000 |
| SMILES | [Cl-].[NH4+] |
|
InChI
InChI=1/ClH.H3N/h1H;1H3
|
|
| Properties | |
| Molecular formula | |
| Molar mass | 0 g mol-1 |
| Appearance | White solid, hygroscopic |
| Odor | Odorless |
| Density | 1.519 g/cm3 |
| Melting point | |
| Boiling point | |
| Sublimes at 337.6 °C ΔsublH |
|
| 244 g/L (−15 °C) 294 g/L (0 °C) 383.0 g/L (25 °C) 454.4 g/L (40 °C) 740.8 g/L (100 °C) |
|
|
Solubility product (Ksp)
|
30.9 (395 g/L) |
| Solubility | Soluble in liquid ammonia, hydrazine, Slightly soluble in acetone Insoluble in diethyl ether, ethyl acetate |
| Solubility in methanol | 32 g/kg (17 °C) 33.5 g/kg (19 °C) 35.4 g/kg (25 °C) |
| Solubility in ethanol | 6 g/L (19 °C) |
| Solubility in glycerol | 97 g/kg |
| Solubility in sulfur dioxide | 0.09 g/kg (0 °C) 0.031 g/kg (25 °C) |
| Solubility in acetic acid | 0.67 g/kg (16.6 °C) |
| Vapor pressure | 133.3 Pa (160.4 °C) 6.5 kPa (250 °C) 33.5 kPa (300 °C) |
| Acidity (pKa) | 9.24 |
| -36.7·10−6 cm3/mol | |
| Refractive index (nD) | 1.642 (20 °C) |
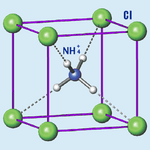
| Structure | |
| Crystal structure | CsCl, cP2 |
| Space group | Pm3m, No. 221 |
| Lattice constant | a = 0.3876 nm |
| Thermochemistry | |
| Std enthalpy of formation ΔfH |
−314.43 kJ/mol |
| Standard molar entropy S |
94.56 J/mol·K |
| Specific heat capacity, C | 84.1 J/mol·K |
| Pharmacology | |
| ATC code | |
| Hazards | |
| NFPA 704 |
|
| Flash point | Non-flammable |
| U.S. Permissible exposure limit (PEL) |
none |
| Related compounds | |
| Other anions | Ammonium fluoride Ammonium bromide Ammonium iodide |
| Other cations | Sodium chloride Potassium chloride Hydroxylammonium chloride |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Ammonium chloride is a chemical compound composed of ammonium and chloride ions. It is a colorless crystalline compound. It is used in soldering. It is also used as an electrolyte in the Leclanche cell, a type of primary cell. Ammonium chloride can be deprotonated by strong bases such as sodium hydroxide to produce ammonia gas.It is sublime compound.
Related pages
Images for kids
See also
 In Spanish: Cloruro de amonio para niños
In Spanish: Cloruro de amonio para niños

All content from Kiddle encyclopedia articles (including the article images and facts) can be freely used under Attribution-ShareAlike license, unless stated otherwise. Cite this article:
Ammonium chloride Facts for Kids. Kiddle Encyclopedia.



